The second LEDs Magazine Lighting for Health and Wellbeing conference included a stellar roster of researchers and human-centric lighting experts, writes MAURY WRIGHT, and some of the more interesting talks focused on characterizing lighting for health and the direct positive and negative impacts of artificial light on health.
LEDs Magazine held its second Lighting for Health and Wellbeing Conference on July 18 in Newport Beach, CA, and the attendees got to hear from the top experts in human-centric lighting (HCL; Fig. 1). Many of the talks focused on the influence of lighting on non-visual systems and the circadian impact of lighting, although the program also included coverage of LED lighting as a way to reduce the occurrence of myopia (watch for an upcoming feature article), continuous disinfection through lighting, and lighting for health in the built environment. Perhaps the most compelling subjects of the day focused on characterizing lighting for health and understanding the direct impacts of light on health. We will focus on several of those presentations here.
FIG. 1. Conference attendees immersed themselves in topics from metrics to circadian impact to applying light for disinfection as well as therapeutic effects.
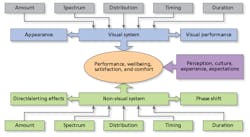
Mariana Figueiro, PhD, director of the Lighting Research Center (LRC) at Rensselaer Polytechnic Institute, opened the day delivering the Keynote Presentation. She posed the question, “What does light affect?” And she showed the diagram in Fig. 2, indicating an impact on visual and non-visual systems in addition to the psychological system.
FIG. 2. Visual, non-visual, and psychological systems are all impacted in different ways by lighting.
Figueiro said the systems on the chart “may be a small part, but it’s the part we know best.” Light could impact far more systems than are known today. “Even within these systems, I think lighting has different characteristics and impacts these systems differently,” said Figueiro.
The point is that it’s very complex and not fully understood at this point — a point echoed by many other speakers on the day. “But I don’t think we should just throw our hands up and say, ‘Oh my god, I can’t do anything,’” said Figueiro. She would go on to describe some steps that she urged practitioners to take now.
Psychological effects
Figueiro quickly moved into the perception of psychological effects on a person and focused in on red light. She said researchers at the LRC had noticed that red had an alerting effect on people. But the researchers assumed they were mistaken, because conventional wisdom holds that blue-rich light drives alertness. “That’s one thing I want you to remember from this talk,” said Figueiro. “It’s not just about blue.”
She referenced the work of Andrew Elliot, psychology professor at the University of Rochester, who has said that red is a danger cue for humans and it triggers a fight-or-flight reaction in people. Research has even shown that humans judge a person wearing a red shirt as being angrier than others wearing, say, blue or white.
Figueiro said the LRC has been looking more at the non-visual physiological effects of light rich with red energy. She said the research shows that red light during the post-lunch dip, or in the middle of the night, can improve alertness and reduce reaction time.
Circadian Stimulus
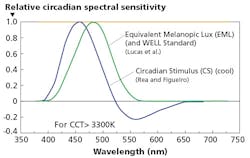
Next, Figueiro began to discuss the LRC’s work on a spectral sensitivity function called Circadian Light or CLA that further leads to the Circadian Stimulus (CS) metric. She said most current thinking on physiological response is that it’s not just melanopsin and the intrinsically photoreceptive retinal ganglion cells (ipRGCs) of the non-visual system that impact physiology. The visual-centric rods and cones play a role as well, although researchers are still working to understand the interaction. At the Lighting for Health and Wellbeing Conference in 2017, Jamie Zeitzer of Stanford University suggested that the visual system may provide some type of gain-control function.
Figueiro’s point, however, is that another popular metric, Equivalent Melanopic Lux (EML), is really tied specifically to the ipRGCs. EML does not account for any impact of the rods and cones. The CLA function has peak sensitivity around 460 nm. Figueiro explained that the model changes based on the spectral power distribution (SPD) and the amount of light, and the LRC has defined a straightforward way to calculate CLA.
Still, CLA alone is not enough for an accurate characterization of the impact of light on a person because Figueiro said it’s not just about spectrum, amount matters also. She said a CLA or EML measure alone would not be easy to evaluate as to threshold of impact or saturation of the physiological system. So the LRC developed the transfer function that uses CLA to derive CS, which characterizes the response by the circadian system.
Limitation of metrics
Figueiro warned of some limitations of using CS and also explained some subtle details of applying it. The work thus far is based on one-hour exposure time to a specific light source. And it only considers melatonin suppression as a measure. But duration matters, and light impacts many physiological systems beyond melatonin level. And she stressed that applying CS requires a strict measure of light at the eye. A measure of horizontal illuminance would not deliver accurate results. Measuring at the eye takes into account the full beam distribution and reflections from surfaces in a space. The LRC has recommendations on CS levels for different times of day and tasks at hand.
Referring to tests based on the CS metric, Figueiro offered some of her conclusions as to what we know. She said light has a direct or acute impact on people and that it doesn’t have to be blue light that is used to increase alertness. For example, you can use red light at night to achieve the impact without changing a person’s circadian phase. With different SPD, you may need to increase the light level to achieve the same CS. Meanwhile, work continues on the impact of duration, but Figueiro said it appears duration beyond two hours seems to have a much lower impact.
Returning to the theme that we don’t know everything, but we should do something, Figueiro concluded, “I think that if we over-design... If we just get bright light during the day, because right now we are under dim light constantly. If we just increase a little bit more… I think we can start making a change in the builtenvironment.”
Characterizing circadian potency
Also focused on the topic of metrics was Martin-Moore-Ede, MD and PhD, founder of Circadian Light and the professor who led the team that discovered the SCN (the body’s master pacemaker) back in 1980 at Harvard. Moore-Ede’s presentation was entitled “Circadian potency: Circadian spectral sensitivity to white light in the workplace.” He began by asking the question, “What is the spectral sensitivity that we need to work with?” He noted that the $120 billion lighting industry needs to know this, and lighting designers and specifiers also need to know.
Moore-Ede almost immediately addressed the aforementioned circadian models in wide use today — CS and EML — and noted a major problem in that the models give conflicting advice to the industry. He referenced a survey conducted in late 2017 by Metropolis Magazine across the architectural design community that found the participants universally confused by the metrics, although 62% said they use CS while 38% said they use EML.
Depicting the CS and EML sensitivity models (Fig. 3), Moore-Ede dug into the science behind the two. CS is based on testing people who have been fully dark adapted, having been in the dark for two hours with dilated pupils. The test subjects are then subjected to relatively-short 30–90-minute monochromatic light exposures. The researchers looked for a transient melatonin event. That test was augmented with modeling of the rods and cones, according to Moore-Ede.
FIG. 3. The Circadian Stimulus and Equivalent Melanopic Lux spectral sensitivity models give conflicting guidance on human-centric lighting.
The EML model is based largely on animal studies, noted Moore-Ede. The metric relies on using genetically-modified cells to produce human melanopsin, and then exposing such cells to very short flashes of monochromatic light at varying wavelengths. The result is determined by measuring calcium response in the cells.
Metric rooted in work conditions
But Moore-Ede said neither was derived under real-world conditions such as he said lit the room in which the conference took place. He characterized such conditions as polychromatic white light in a room with freely moving people, with intensity in the 200–1000-lx range, and long exposure duration. And he said the existing metrics don’t have a strong correlation to human health.
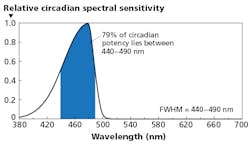
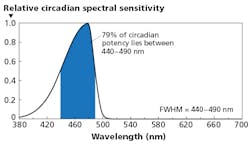
A National Institutes of Health (NIH) Small Business Innovation Research (SBIR) grant has allowed Moore-Ede to study his Circadian Potency metric in a setting equivalent to typical workplace conditions. The study considered polychromatic lighting with varying spectra, all at 540 lx. And to remove any preconceived bias from the results, Moore-Ede hired a mathematician with no real prior knowledge of lighting and spectral sensitivity to perform the data analysis, determining best-fit peak sensitivity and the shape of the left and right sides of the curve with no requirement that the result be symmetrically Gaussian.
Ultimately, the research yielded the Circadian Potency spectral sensitivity curve depicted in Fig. 4. The peak is in the blue region and 79% of the calculated area under the curve (AUC) is in the blue region. Moreover, that shaded region falls within what Moore-Ede said is the steady-state response tied to long exposure, whereas the shorter and longer wavelengths to either side are more transient effects.
FIG. 4. Circadian Light’s proposed Circadian Potency sensitivity model concentrates on response to steady-state stimulus.
You calculate the Circadian Potency metric by comparing the AUC of a light source in that 440–490-nm region and dividing it by the AUC over the entire human visible range. Such data could easily be derived by handheld spectrometers. And light could be characterized and specified based on target Circadian Potency measurements for day and night.
Closing Plenary
The Closing Plenary presentation was made by David Blask, PhD and MD, from the Tulane University School of Medicine. Blask performs research in the Department of Structural and Cellular Biology in the Tulane Cancer Center. He downplayed his accomplishments, saying that he had been a bench scientist working in the circadian aspects of cancer for 30–40 years, implying a somewhat mundane work history. He said that back when he started, “No one was working in the area, especially if you were working on melatonin. But the thing seems to have blossomed, so I’m glad we stuck with it.”
Stealing a theme from a famous Spaghetti Western movie and flipping that theme, Blask’s presentation was entitled “Light, circadian melatonin and cancer: The ugly, the bad, and the good.” Blask recounted, “Light is in fact a double-edged sword with regard to cancer.” The conference and the prevailing wisdom in the human-centric lighting area is that blue-rich light is healthy early in the day, yet light at night can be detrimental to the circadian system. Blask said, “Well, cancer is a disease process that is very responsive to these kinds of changes, induced by light’s ability to modify the circadian system, either in a good way or in a bad way, we think by modulation of the melatonin signal.” He reminded that melatonin is both a circadian signal and an anti-cancer hormone that has direct anti-cancer effects.
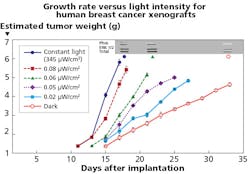
Starting with what he called “the ugly,” Blask said dim light at night (dLAN) can break the circadian clock. Blask’s research is based on using xenografts of human cancer implanted in mice or rats. He said in normal conditions, tumor cells grow more rapidly during the day and more slowly at night when melatonin is present, but that dLAN was the ugly. The characterization is based on tests where rats were subjected to different levels of polychromatic white light at night.
The control conditions were 12 hours of light and 12 hours of darkness. The tests ramped the nighttime light levels the rats were subjected to up to the point that the melatonin level saturated. The graph in Fig. 5 shows that in the control dark-at-night condition, the tumors grow but at a relatively slow rate. As light levels increase, the growth rates accelerate significantly. Blask said growth of the biomass or tumor is fueled by fatty acids and glucose, but the presence of melatonin blocks those fuels.
FIG. 5. Increasing levels of light at night resulted in faster-growing tumors in rats.
Blask went on to show graphs of the biochemistry that would indicate in normal light–dark cycles the entire process is rhythmic. But even exposure to dim levels of 0.2 lx/cm2 can lead to elimination of the rhythmic patterns and 24-hour-per-day growth. The result is two- or threefold greater levels of tumor growth. He did say rats are far more sensitive to light than humans, implying that such dim exposure would not have the same impact on humans.
Relating research to humans
Blask lamented that you can’t easily perform the needed long-term testing on humans to replicate the work with rats. But Blask’s team has worked to approximate human testing with a team from Thomas Jefferson University led by George (Bud) Brainard, PhD. Brainard’s team collected blood samples from female volunteers during the day, during the night, and after exposure to dLAN. Those samples were shipped to Blask’s team where they had implanted human breast cancer xenografts into rats and developed a way to pump the blood samples stored in an external source directly to the tumors — what Blask termed a human-to-human process.
The research delivered the expected results with melatonin suppression and tumor growth during the day, retarded growth at night, and growth near daytime levels after dLAN. Blask said, “This really defined for the first time that indeed the melatonin signal in humans was a circadian anti-cancer signal that would protect you from the progression of developing clinical cancer.” Brainard and Blask have done very similar research and documented similar results with prostate cancer in males.
Having documented the impact of dLAN on males and females, Blask said the team wondered if there was a spectral aspect to the light exposure. Would perhaps red-rich light have less impact suppressing melatonin than blue-rich light? And testing based on a lengthy dark exposure to establish normal melatonin levels and subsequent exposure to red light did not suppress the level, whereas blue exposure did suppressmelatonin.
The bad and good
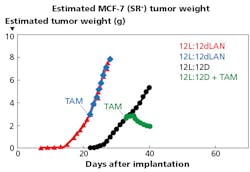
Next, Blask moved on to what he called “the bad.” He said the team considered the impact of treating breast cancer implanted in rats with tamoxifen — a first-line hormonal therapy for breast cancer. The idea was to compare the response to tamoxifen treatment in rats with an intact melatonin signal with others that had melatoninsuppressed.
When tamoxifen was introduced after a tumor had begun to grow rapidly in a population exposed to dLAN, the growth rate continued to escalate. When it was introduced to a population that was subjected to a normal 12-hour dark cycle, the tumor growth stagnated and then regressed (Fig. 6). Blask said, “Light at night not only impacts the intrinsic activity and ability of these tumors to grow and metabolize and progress to a potentially clinical cancer state, but this light at night under these experimental conditions leads to tamoxifen resistance.” Blask said the team has repeated the process with other chemotherapies and documented the same exact response.
FIG. 6. The hormonal breast cancer therapy tamoxifen (TAM) was rendered ineffective when rats were exposed to light at night.
Blask then said, “Now I want to end with the good, or the potential good.” Turning from a discussion of dLAN, Blask focused on the use of bright blue-enriched LED light during the day. “We like to think of blue light as reinforcing the circadian system,” said Blask. The research showed that daytime exposure to standard polychromatic white light, such as a fluorescent source, and a blue-enriched source, both at identical photon densities, resulted in a nighttime surge in melatonin levels by a six-to-seven-fold factor for the blue-rich light. Moreover, the duration of the melatonin presence is expanded by two to three hours.
Blask asked, “Does this have consequences for tumor growth?” And immediately answered, “It sure does... It’s as if we have given these animals a melatoninsupplement.”
Blask said the team performed research using mice implanted with liver cancer to characterize the impact of what they call bLAD (blue light at daytime). The mice were treated with a state-of-the-art chemotherapy drug used to treat liver cancer, and with a normal day–night cycle, the tumors were shown to regress. But mice also exposed to bLAD exhibited tumor regression at double the rate of those that were studied under the standard light cycle.
Next steps, according to Blask, will be to develop human applications using tunable circadian lighting to modify the melatonin anti-cancer signal to suppress tumor growth and metastasis. And the team wants to use that tunable lighting to increase sensitivity to cancertherapies.
Of course, there will be many less-dramatic uses than cancer for human-centric lighting. Continuing investigation of the science will generally improve sleep patterns and productivity. And the LRC’s Figueiro has research suggesting that tunable lighting can improve the lives of dementia patients. As manufacturers understand the technology and designers learn to broadly specify it, lighting for health and wellbeing has a bright opportunity and offers great profit potential for the solid-state lighting (SSL) industry.