“Sunlight is the best disinfectant,” the old adage goes, but it is the unseen ultraviolet (UV) component of sunlight that really gets the job done. Early discoveries of UV light’s disinfecting power 150 years ago led to the development of modern ultraviolet germicidal irradiation (UVGI) systems that combat any number of dangerous pathogens. For pandemics of the past, this disinfection technique was not available; however, diseases traveled the globe more slowly, limited by the fastest mode of travel at the time. Today with modern air travel, we have seen the COVID-19 pandemic traverse the globe in a matter of days. It has been roughly 100 years between the Spanish Flu and COVID-19 pandemics; however, outbreaks are far more common, as we have experienced several near misses in the last 20 years, such as the swine flu, bird flu, and SARS.
Another area driving the need for UVGI is the growing development of antimicrobial-resistant (AR) pathogens. All microorganisms can develop a form of AR, with the most common form being antibiotic resistance. Antibiotic-resistant pathogens, such as methicillin-resistant Staphylococcus aureus (MRSA), render typical antibiotic treatments less effective because the pathogen has acquired a chemical resistance to the drug. UVGI combats these organisms in the environment without producing a resistant strain. Continuing innovations in UV light sources will help increase the safe application of the technology in the ever-widening UV disinfection market. This article explores the various UV sources for germicidal disinfection and provides some background on which factors are most critical in determining a UVGI solution’s effectiveness.
Importance of UV wavelength and dose
Ultraviolet energy is a general term for electromagnetic radiation for wavelengths spanning from 100–400 nm, and categorized into four bands:
- UV-V (100–200 nm) — Light rays can travel only in a vacuum
- UV-C (200–280 nm) — Most harmful to DNA/RNA
- UV-B (280–320 nm) — Typically used in curing and tanning applications
- UV-A (320–400 nm) — Most commonly used in LED curing applications
Editor’s note: IEC standards consider vacuum UV (UV-V) a sub-band of UV-C.
UV light does not kill organisms directly. Instead, UV-C photons (those between 200–280 nm, and especially around 260 nm) form pyrimidine dimers in the base pairs of DNA and RNA that prevent pathogenic organisms from reproducing. UV-B radiation (280–320 nm) also works but to a lesser degree.
Most organisms have a genetic self-repair mechanism, so UVGI systems must deliver a sufficient UV dose to overwhelm their defenses and prevent replication. “UV dose” can be defined as a product of light energy intensity and exposure time (UV irradiance in W/cm2 × time in seconds), usually measured in units of J/cm2. UV-C for disinfection is an effective method for deactivating pathogenic reproduction, where dose effectiveness is measured in terms of the reduction of infectious organisms in logarithmic units. A reduction of one million is “6-log,” while a reduction of one thousand is “3-log,” and so forth. A recent study at Henry Ford Hospital in Detroit, MI, for example, showed that a dose of 254-nm light of 1 J/cm2 effectively reduced H1N1 flu viruses by a factor of 1000 (or 3-log) on exposed surfaces of medical respirator masks.
UVGI systems work only in line-of-sight disinfection of transparent liquids and hard surfaces (e.g., tables/counters, walls, bed rails, keyboard surfaces, etc.). Shadowing, or areas without direct line-of-sight exposure to the UV-C source, will not be disinfected, so use of multiple light sources, reflective surfaces, and/or motion-enabled sources should be considered. While UVGI is used for disinfecting pathogens from air in HVAC systems or in the upper levels of rooms in medical or educational facilities, it is less effective for use on porous materials like carpets or cloth.
Lamp-based UV-C sources and design considerations
UVGI system designers face challenges in working with UV-C light as most materials do not reflect or transmit UV-C energy very well. UV-friendly lenses made from fused silica, quartz, and other specialty glasses must be used to direct UV-C light. Reflective elements include Teflon, PTFE (polytetrafluoroethylene), and UV-enhanced aluminum. UV-C light can severely damage skin and eyes, so attention to human safety measures is crucial. UVGI systems usually operate where there is no chance of contact with living tissue, but technicians who build and use these systems require protection.
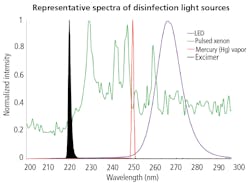
At the heart of any UVGI system lies a UV-C light source. Common UV light sources used for disinfection include low-pressure mercury-vapor lamps, pulsed-xenon lamps, excimer lamps, and UV light-emitting diodes (LEDs).
Peter Cooper Hewitt patented the first mass-produced mercury-vapor lamps in 1901, and this technology is still in use today. Low-pressure mercury (Hg) vapor lamps resemble everyday fluorescent lamps without an internal phosphor coating to convert UV energy into visible light. Rather than using borosilicate glass, which absorbs UV, they incorporate fused quartz or titanium (Ti)-doped quartz to block the ozone-producing 185-nm emission from low-pressure Hg.
Because of their relatively long life and high electrical-to-optical conversion efficiency near 30%, low-pressure Hg lamps remain a cost-effective solution for many UVGI systems. However, Hg lamps are less physically robust than other solutions and pose a risk of toxicity when broken or discarded. They also have difficulty operating effectively in environments below 40°F (4.4°C) as colder temperatures hinder the liquid Hg from vaporizing.
Alternatively, pulsed-xenon lamps deliver broadband UV-C emission from 200–300 nm, without mercury or other toxins. These lamps generate little heat but deliver high irradiance levels of 1 kW/cm2 or more, which makes them ideal for disinfecting larger areas when humans are not present. However, pulsed-xenon lamps may require filtration to remove unwanted wavelengths, and also use relatively expensive support electronics that can increase overall system costs.
Excimer lamps offer yet another UVGI light-source alternative. They incorporate diatomic molecules like potassium bromide (KBr) or potassium chloride (KCl) that are stable in higher electronic states but relax to unstable status at lower electronic states, emitting UV-C photons at 207 or 222 nm. Excimer lamps typically feature no metallic electrodes, so they are more mechanically robust than Hg or pulsed-xenon sources. The outer layer of human skin, comprising dead cells, readily absorbs shorter UV wavelengths generated by excimer lamps. This effect has led to new research to determine whether 222-nm energy might be germicidally effective and also safe for prolonged human exposure.
UV-C LED sources for UVGI
Advances in semiconductor design have yielded LEDs based on aluminum gallium nitride (AlGaN) that emit UV-C light near 214, 260, and 275 nm. UV-C LEDs are compact, most easily integrated into systems for point-of-use UV disinfection applications, and turn on almost instantaneously with no power-up delay. This makes them ideal for sporadic on/off operation only in the absence of human bodies. LEDs also operate at relatively low voltages for easier integration into compact or handheld devices powered by batteries or solar cells.
The cost per unit of power for UV-C LEDs remains higher than lamp-based sources, but UV-C LEDs deliver enough power for most dosage requirements and applications. A 2016 study at the Universities of Colorado and North Carolina showed 260-nm LEDs were at least as effective as low-pressure Hg systems in deactivating E. coli and bacterial spores. Work at a leading UV-C LED manufacturer demonstrated successful inactivation of the SARS-CoV-2 virus, while other research successfully inactivated other human coronaviruses. More recently, researchers at the University of Miyazaki in Japan showed rapid deactivation of in vitro SARS-CoV-2 using doses as low as 37.5 mJ/cm2 with LEDs near 280 nm.
Processing improvements to increase LED output power are in development. New crystal-growth techniques reduce losses caused by dislocations near the interface between the AlN-based semiconductor and its sapphire substrate. University of California at Santa Barbara researchers also demonstrated improved performance using silicon carbide (SiC) instead of sapphire. Design improvements promise to more efficiently direct light out of the semiconductor. In the same way that blue LEDs’ performance for color displays improved by an order of magnitude through the 1990s and early 2000s, advances in UV-C LED performance are expected to continue making this technology more attractive for the next generation of UVGI systems.
Selecting a UVGI source
The question as to which light source is ideal remains. Although we cannot answer that question directly, we can provide insight into a solution for a given application (see the nearby table for feature comparisons). In general, if your solution requires 10W or greater of UV-C optical power, a mercury-vapor or pulsed-xenon light source will offer the best price versus performance. Although the wall-plug efficiency of UV-C LED is increasing, the current efficiency and low cost of mercury-vapor and pulsed-xenon light sources cannot be matched. If your solution requires less than 10W of UV-C optical power, then the advantage tilts toward UV-C LEDs. Although the cost per unit of optical energy is higher, the small footprint, robust design, and low voltage enable easier integration. Although excimer lamps offer the possibility of prolonged UV exposure at 222 nm and below, their cost and limited lifetimes limit their range of applications. Until either of these factors change, this source will likely be limited to hospital-based applications where the benefit far outweighs the device cost.
Conclusion
Ultraviolet light has long served as a powerful tool to combat pathogens. New interest in the germicidal power of UV light is driving continuous innovations in UV-C light sources that promise to make UVGI a more prominent weapon in the fight against new and more dangerous pathogens such as SARS-CoV-2 and antibiotic-resistant bacteria in a wide range of settings.
Get to know our experts
PAMELA LEE is senior product manager and MARVIN RUFFIN is UV business development manager at Excelitas Technologies, a manufacturer of photonics technologies used in applications from medical, sciences, and safety to automotive, aerospace, and defense, and more.
For up-to-the-minute LED and SSL updates, why not follow us on Twitter? You’ll find curated content and commentary, as well as information on industry events, webcasts, and surveys on our LinkedIn Company Page and our Facebook page.